Rev Bras Oftalmol.2025;84:e0084
Cost-utility analysis of latanoprostene bunod eye drops in glaucoma treatment from the perspective of the Brazilian Unified Health System
DOI: 10.37039/1982.8551.20250084
ABSTRACT
Objective:
To evaluate the cost-utility of latanoprostene bunod 0.024% as first-line therapy compared to the strategy recommended by the Clinical Protocol and Therapeutic Guidelines for primary open-angle glaucoma of the Brazilian Unified Health System.
Methods:
A cost-utility analysis was conducted using a hypothetical cohort and a Markov model. In the base-case scenario, the treatment strategy recommended by the Clinical Protocol and Therapeutic Guidelines was adopted, initiating therapy with timolol 0.5%. If the target intraocular pressure was not achieved, dorzolamide 2% and prostaglandin analogue were added. The comparator scenario considered latanoprostene bunod 0.024% as the initial treatment option, followed, when required, by the sequential addition of timolol 0.5% and dorzolamide 2%.
Results:
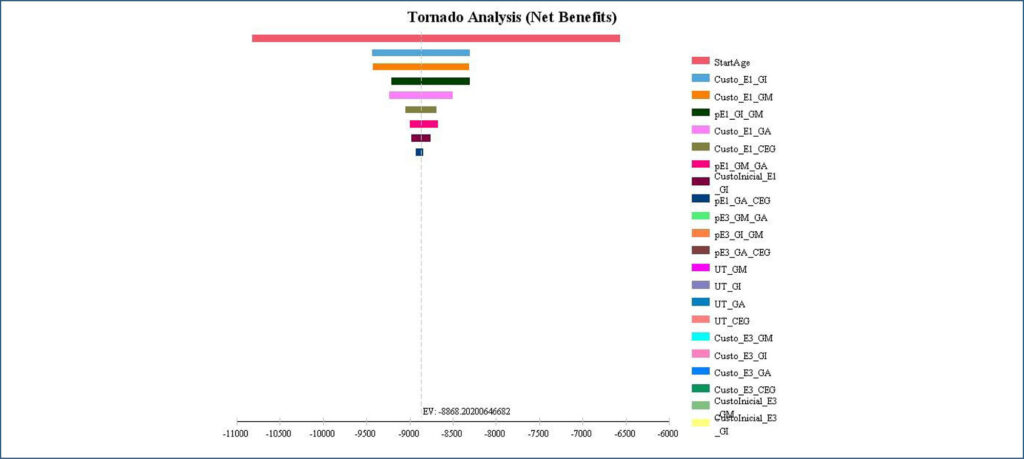
The model analysis showed a cost-utility ratio of BRL 31,244.98 per Quality-Adjusted Life-Years for the latanoprostene bunod 0.024% strategy compared to the Brazilian Unified Health System strategy. Age at model entry was the parameter with the greatest impact on cost-utility ratio, resulting in a range from BRL 24,953.82 to BRL 50,284.30 per Quality-Adjusted Life-Years.
Conclusion:
Both strategies provide gains in quality of life, and the incorporation of latanoprostene bunod 0.024% as a first-line medication for clinical management of primary open-angle glaucoma is considered cost-effective from the Brazilian Unified Health System perspective.